Medical Grade Power Adapter: Ensuring Safety and Reliability in Healthcare
A medical grade power adapter is required for healthcare equipment that requires dependability and exacting safety regulations. Constructed under various rules applicable to medical equipment, the adapters ensures patient’s safety and user’s safety. Medical grade adapters apply quality standards, which standard power adapters don’t apply. They additionally test the adapter with IEC60601-1, UL, CE and other international standards.
Medical personnel use these adapters to power important devices like patient monitors, infusion pumps, and diagnostic devices. A failure or defect may have dire consequences, necessity for high caliber components.
In this article, we will go over specifications, certifications, manufacturing standards, and other considerations for choosing the best medical grade power adapter. Hospitals, clinics and medical device manufacturers who want to ensure safety, compliance and performance need to know this.
Medical Grade Power Adapters Explained
This has a high reliability and reduces risk failures during high-stakes procedures. It can withstand environment heat, moisture and vibration.
Relevance of Certification
| Feature | Medical Grade Adapter | Standard Adapter |
| Electrical Isolation | Yes | No |
| Certification Compliance | IEC60601-1, UL, CE | Limited or none |
| Safety Assurance | High | Moderate |
| Reliability | Very high | Variable |
Medical grade adapters ensure devices work safely during constant use in medical applications, providing peace of mind to users.
Manufacturing Norms and Quality Assurance
Development Method
Producing medical grade power adapters requires meticulous engineering and quality check during every production stage.
Assessing durability for long-term reliability
Manufacturers of medical grade adapters use an ISO certified process to maintain consistency in volume.
Example analysis
- A power adapter was needed for patient monitors.
- Adapters must meet stringent safety and dependability requirements.
- Partnered with a manufacturer of certified medical-grade adapters.
The result of this was zero device failures, regulation compliance and an increase in staff confidence.
Reliability Comparison
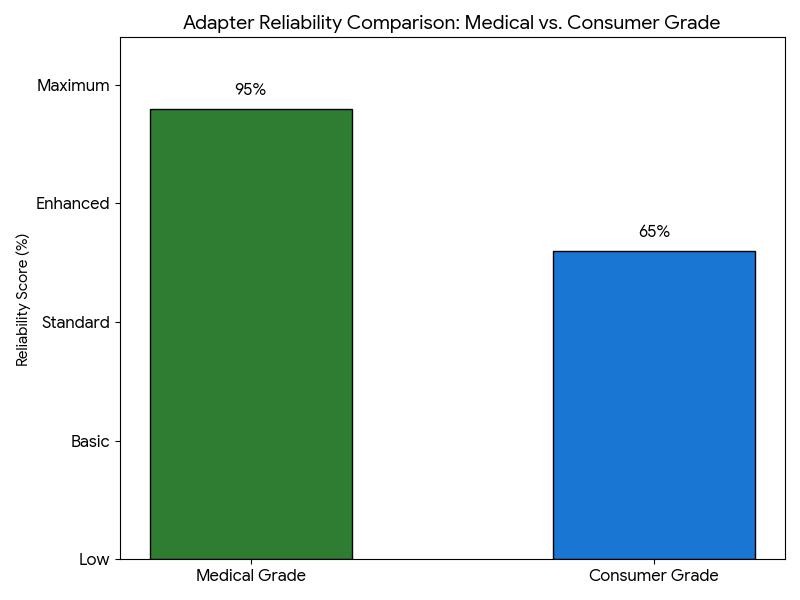 Medical Grade adapters assure high reliability scores and operate without failure in life-critical configurations.
Medical Grade adapters assure high reliability scores and operate without failure in life-critical configurations.- Medical adapters are certified for healthcare safety standards. These include IEC 60601-1. This is a medical equipment certification. This ensures safety for patient and operator.
- Consumer Grade adapters, being medium reliable, are suitable for standard electronics, but their shielding and long-term durability are not up to the mark of products built for medical electronic equipment due to their lower cost.
Tips for Choosing the Best Travel Adapter
Guidelines in Steps
- Examine Certifications: Verify IEC60601-1, UL, CE compliance.
- Evaluate Electrical Specs: Voltage level, current, Isolation level
- Checks how resistant heat, vibration, moisture is our product.
Further Considerations
- Producer familiarity with medicine items.
- Clear documentation and technical assistance.
- Options that can be customized.
- Guarantee and after-sales assist.
The technical requirements and other parameters to choose the right medical grade power adapter are assessed.
Final thought
A power adapter designed for medical use is an integral requirement for the safety and compliance of any medical device. Certified adapters prevent the electrical hazards and device malfunctions from counterfeit ones.
Safety, regulations, and patient care are key benefits of investing in quality neural adapters. Shenzhen Sunray Technology Limited provides dependable power solutions that enhance equipment performance and reduce operational hazards for hospitals and device manufacturers.
Selecting a certified power adapter from a trusted healthcare manufacturer can ensure the device will function as designed, leading to better patient outcomes.
Questions readers usually ask next
Medical grade power adapter ensures safety reliability and stable performance for medical devices meeting strict healthcare standards.
Planning points worth checking early
These notes are here to make the next comparison or sourcing step more concrete.
What this clarifies
Use the article to narrow the next technical or sourcing question instead of staying broad.
What to compare
Check the closest related product family, requirement, or compliance route next.
When to ask
Move into contact or quotation once the issue is specific enough to affect feasibility or timing.
Keep comparing options
These references help visitors move into the most relevant next comparison or conversation.
Medical Grade Power Adapter Manufacturer
See how medical grade power adapter manufacturer supports the next product, sourcing, or factory-planning decision.
Certifications & Compliance
Review how approvals, labels, destination-market rules, and paperwork influence commercial readiness.
Request a Quote
Share the project scope, target markets, and volume expectations to move into a practical quotation discussion.
Contact Us
Reach the team directly for technical questions, sampling requests, and sourcing follow-up.
Questions readers often ask next
Each answer is written to help the next decision feel more concrete.
Why do readers usually look for this topic before sourcing decisions?
Because Medical Grade Power Adapter usually affects specification choices, supplier conversations, or the way risks are compared before a program moves into sampling or volume planning.
What detail should be clarified next after reading about Medical Grade Power Adapter?
The next useful step is usually to define the requirement that will affect feasibility most, such as output level, destination market, safety path, connector format, or packaging direction.
Is this topic more useful for engineering, sourcing, or product teams?
It is often useful across all three because the strongest launch decisions usually come from aligning technical fit, commercial requirements, and timeline expectations instead of treating them separately.
When should someone move from reading to requesting a quote or technical review?
That usually makes sense once the key questions are focused enough to influence feasibility, approval timing, sampling scope, or pricing, even if every detail is not finalized yet.
Need help turning this reading into a product decision?
Share the application, target output, and destination markets if the article has narrowed the next discussion enough to review practical options.